Dr. Gale will serve as Chair, Department of Microbiology and Immunology (DMI) at the University of Minnesota School of Medicine, and Director of the University of Minnesota Institute on Infectious Diseases (UMIID).
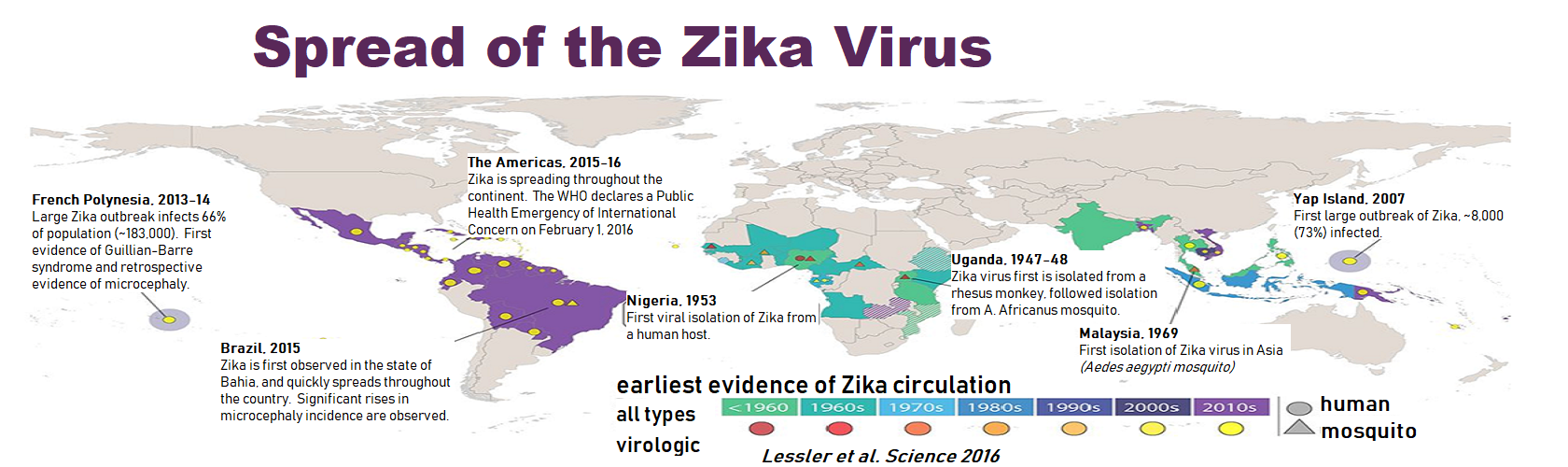
Disruption of myelin structure and oligodendrocyte maturation in congenital Zika infection
New research from the Gale lab reveals that maternal to fetal prenatal exposure to Zika virus leads to profound disruption of fetal myelin, myelin unwrapping from neurons, and extensive suppression in gene expression for key components of oligodendrocyte maturation and myelin production. Because myelin is critical for cortical development, Zika virus-related perturbations in fetal brain myelination could render long-term consequences on childhood neurodevelopment resulting from Zika virus infection during pregnancy.
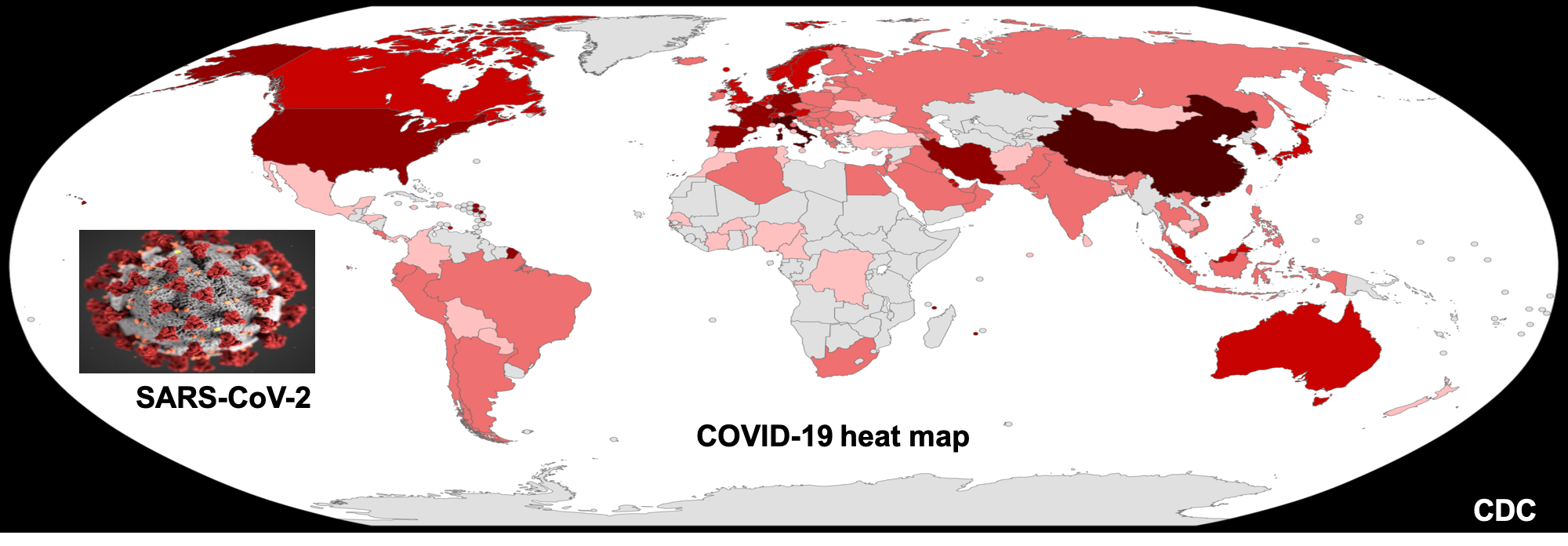
Duration of viral infectiousness and correlation with symptoms and diagnostic testing in non-hospitalized adults during acute SARS-CoV-2 infection: A longitudinal cohort study
In a collaborative study we evaluated viral shedding, and we isolated and sequenced virus across time, comparing outcomes with current diagnostic testing and clinical features of infection and recovery. Remarkably, some people are viral RNA+ for up to 30 days following COVID symptom onset.
Read the study: https://pubmed.ncbi.nlm.nih.gov/36913789/
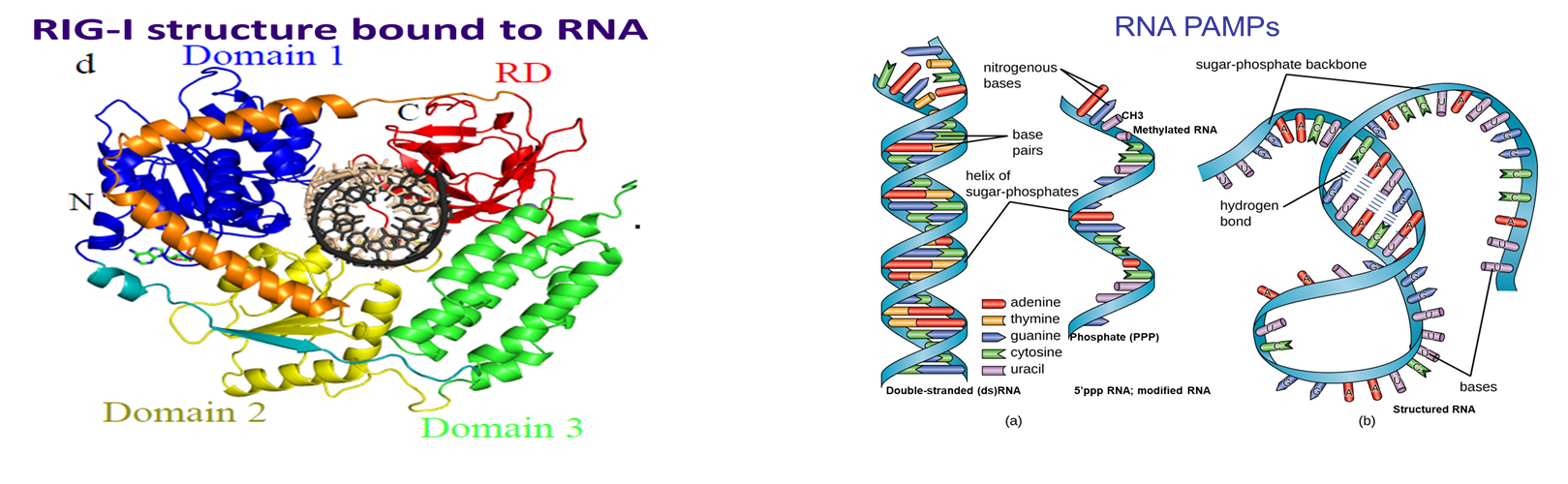
A Small Molecule RIG-I Agonist Serves as an Adjuvant to Induce Broad Multifaceted Influenza Virus Vaccine Immunity
Targeting RIG-I with small molecules for induction of innate immunity enhances influenza A virus vaccine immunity including heterologous immunity against multiple viral HA types. This study reveals RIG-I as a valid drug target for strategies to effectively enhance broad spectrum antiviral immunity against virus infection.
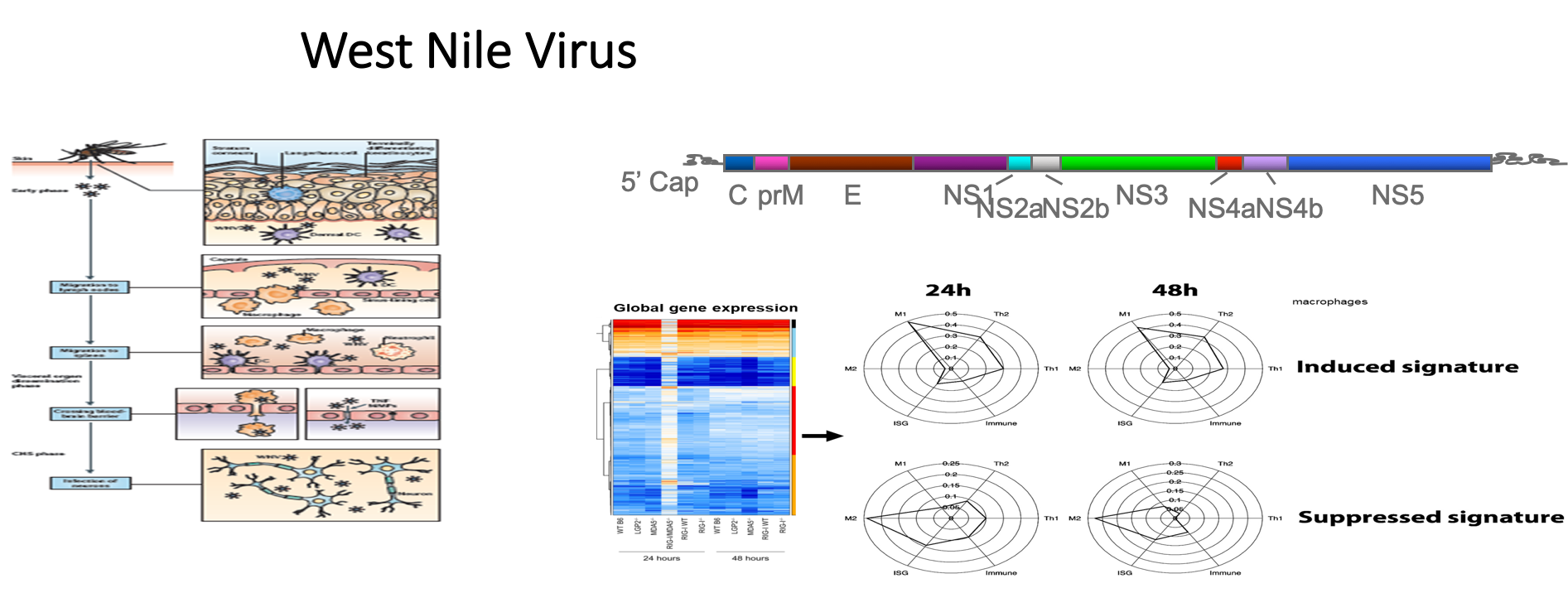
Mouse model of SARS-CoV-2 infection reveals innate immune and inflammatory response differences based on age and gender that accurately model human COVID-19.
In a study led by Dr. Michael Davis, the Gale lab revealed innate immunity as an essential component of disease control and recovery from SARS-CoV-2 infection.
Read the study: https://www.mdpi.com/2076-393X/11/1/47
Innate immune regulation in HIV latency models
A study led by Dr. Rebecca Olson, the Gale lab shows that HIV latency and viral suppression each link with cell-intrinsic defects in specific innate immune gene expression. We identify a set of innate immune effector genes as candidate viral latency restriction factors whose expression and function could possibly mitigate establishing latent HIV infection.
Career Opportunity at IDCRC Laboratory Operations Unit (LOU)
New job opportunity: Are you interested in advancing clinical research in infectious disease vaccines and therapeutics? The Laboratory Operations Unit of the IDCRC is hiring!
The Gale lab has been named on the annual Highly Cited Researchers 2022 list from Clarivate.
The Gale lab has been named on the annual Highly Cited Researchers each year since 2014, and has made this list again for 2022 from Clarivate. This ranking places our group among the top 1% of all cited research teams in the World across the fields of innate immunity, microbiology, and infectious disease. The Gale lab is joined by additional research groups around the world as leaders in their respective fields.
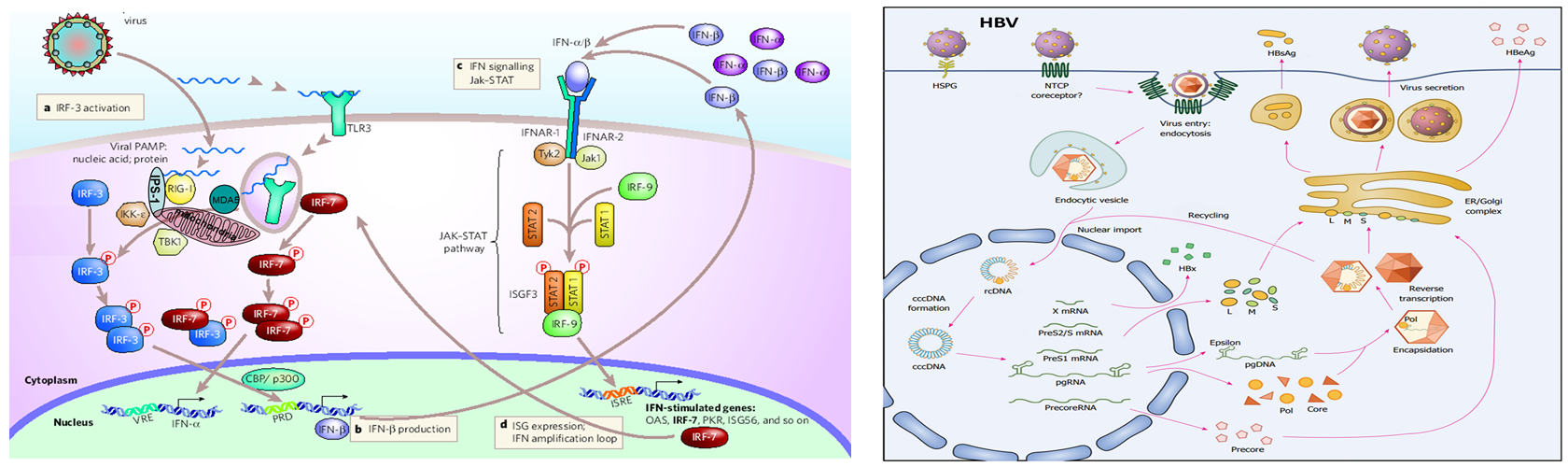
How the Gale Lab “RIG”s innate immune responses against hepatitis B
From the Gale Lab, Pathogen Associated Malignancies Program, Cancer Consortium.
https://www.fredhutch.org/en/news/spotlight/2022/02/ccg-lee-iscience.html
Gale lab is component of USAID virus discovery program
UW joins USAID’s $125M project to detect emerging viruses with pandemic potential.
Check out the USAID’s media release.
Pagination